Research Security Program
Protecting Open Global Collaboration
Why is Research Security Important?
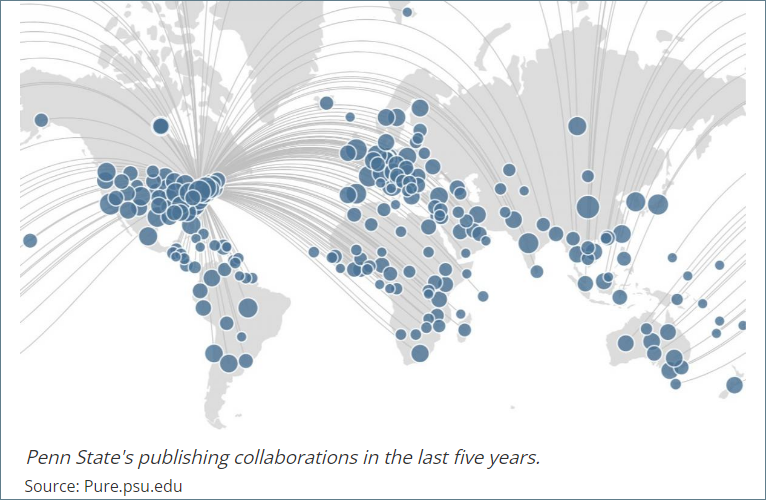
Open scientific and scholarly collaboration between scholars from all over the world is one of the cornerstones of innovation and technological advancement, and Penn State remains committed to fostering such collaboration. While the vast majority of international affiliations do not present a security risk, the Federal government has increased its attention to research in foreign countries. There have been cases at some universities in the United States where researchers have violated laws and policies aimed at preventing lapses in research security. In response to this, Penn State is establishing a Research Security Program.
What is Research Security?
Research security is a broad term that refers primarily to national security concerns surrounding research involving certain types of sensitive data, intellectual property, export controlled information, and other risks. An area which the federal government has focused on as critical to protecting the security of U.S. research is the disclosure of conflicts of interest and commitment, including relationships or affiliations with foreign entities, and research support provided by those entities.
Regulagory Background
Read more about the regulatory background as it relates to research security.
Penn State's Research Security Program
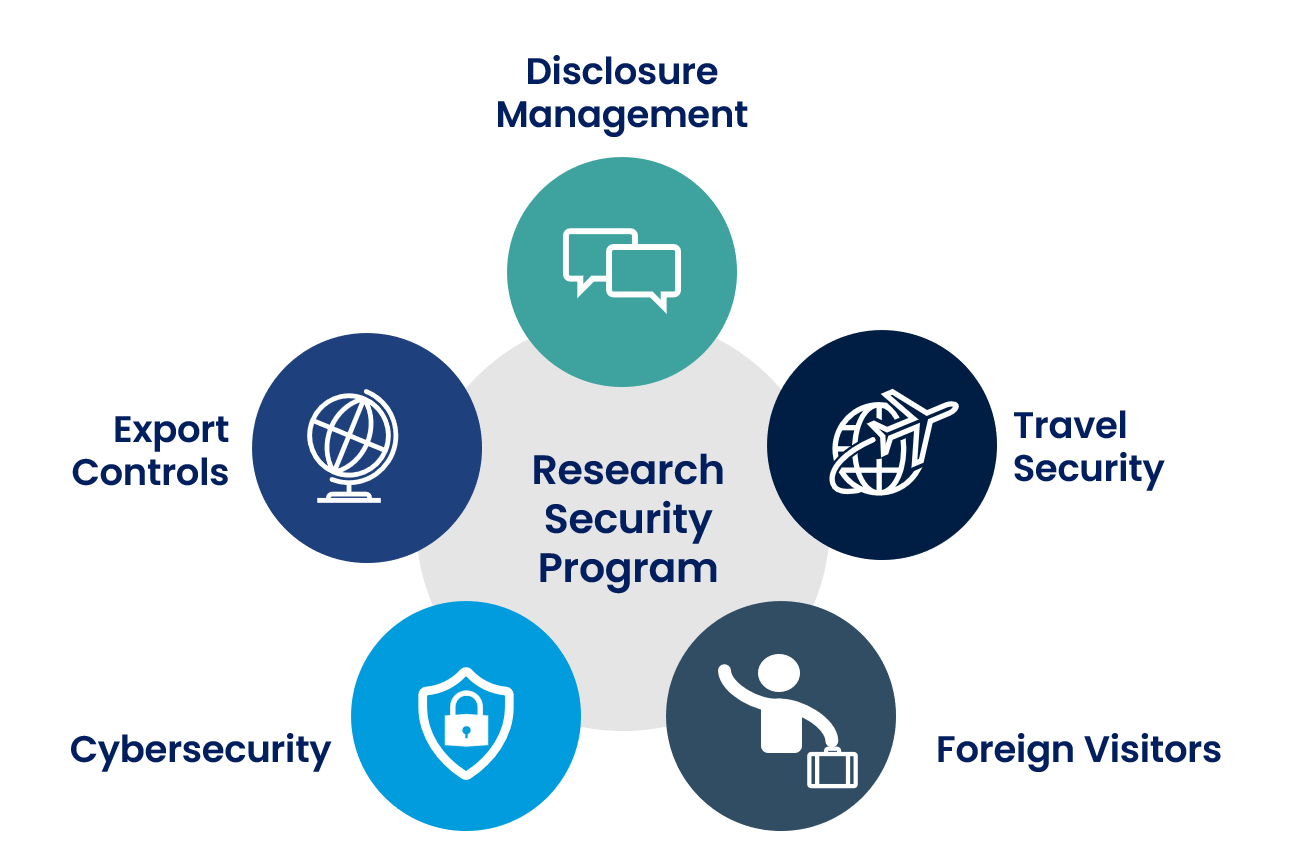
Below are the components of Penn State’s research security program, which is an interdisciplinary collaborative effort among various offices at the University. This program is a central point of contact for research security at Penn State. Please email research-security-orp@psu.edu with any questions.
Cybersecurity is an expansive and complex area and its scope extends well beyond research. For purposes of research security, the following resources are helpful starting points.
Research taking place under government contracts may be subject to additional security controls. For assistance with this type of research, please email the Office of Information Security (OIS) at security@psu.edu.
Digital Persistent Identifiers (DPIs) and Digital CVs make it easier for researchers to create and maintain a complete and up to date record of their publications and achievements in one location, thereby streamlining the grant proposal process. In addition, some federal agencies are now requiring investigators to register with a DPI service, such as ORCID, and submit biosketches and current & pending support via SciENcv. See the following University Libraries webpages for step-by-step guides that walk you through creating an ORCID ID, adding records to your profile from the PSU Researcher Metadata Database, and using SciENcv to create a biosketch:
As indicated above, disclosure and transparency surrounding relationships with, and research support from, foreign entities have been identified by the federal government as key to managing and protecting the security of our research. Researchers are expected to disclose outside financial interests and Outside Professional Activities through the Office for Research Protections (ORP). Similarly, researchers are required to disclose Other Support or Current & Pending Support in their grant and contract applications. The ORP and the Office of Sponsored Programs (OSP) work together to support the disclosure process and are available to answer questions as needed. See the following pages for more information and contacts.
- Conflict of Interest (COI)
- Conflict of Commitment (COC)
- Other Support/Current & Pending Support
- Resource: See the Faculty Outside Activities and Disclosure Guide for helpful information on what activities require disclosure, prior approval, or further review under polices RP06 and AC80.
The Export Compliance Officer maintains responsibility for establishing, implementing, and enforcing University-wide export compliance policies, procedures, and guidelines designed to meet or exceed the requirements of the various federal laws governing the export of goods, technology, and information, including compliance with ITAR, EAR, and OFAC regulations.
A process for reviewing visitors to Penn State is part of the comprehensive approach to research security outlined in NSPM-33. The Visiting Scholars process is composed of reviews by a number of University offices to ensure that our esteemed visitors comply with regulatory requirements.
NOTE: Hosts of Visiting Scholars should be aware that effort from Visiting Scholars who have support from their home institutions may need to be disclosed as Other Support.
- The U.S. Federal government has placed restrictions on researchers participating in Malign Foreign Talent Recruitment Programs. Review RSP's webpage on Foreign Talent Recruitment Programs for more information.
- Get started with building international relationships while staying incompliance with this helpful resource: Building Successful International Research Collaborations
Faculty, staff and students traveling for University purposes (regardless of the funding source) must record international travel in the Travel Safety Network database at least 30 days prior to departure as outlined in the University’s International Travel Requirements Policy TR01. This includes travel for international research, conferences, and internships.
For more information and assistance with planning and conducting international travel, please visit Global Program’s website.
