Continuing Review
According to federal regulations, the IRB must conduct Continuing Review of previously approved research at intervals appropriate to the degree of risk, but not less than once per year [45 CFR 46.109(e) (DHHS) and 21 CFR 56.109(f) (FDA)]. The revised Common Rule (2018 Requirements) outlines circumstances where Continuing Review is not required unless otherwise determined by the IRB. If your study requires Continuing Review, this will be outlined in the study approval letter and the PI will receive reminders from CATS IRB about submitting the Continuing Review. These reviews must be submitted to and reviewed by the IRB prior to the approval end date for the study.
Failure to submit the required form before the deadline moves the study to a lapsed state and requires all research activities to cease until the Continuing Review is complete. If a study is in a lapsed state for 45 days, the IRB office will automatically close the study administratively and the study cannot be re-activated at that time.
Researchers are strongly encouraged to submit Continuing Review several weeks prior to the deadline.
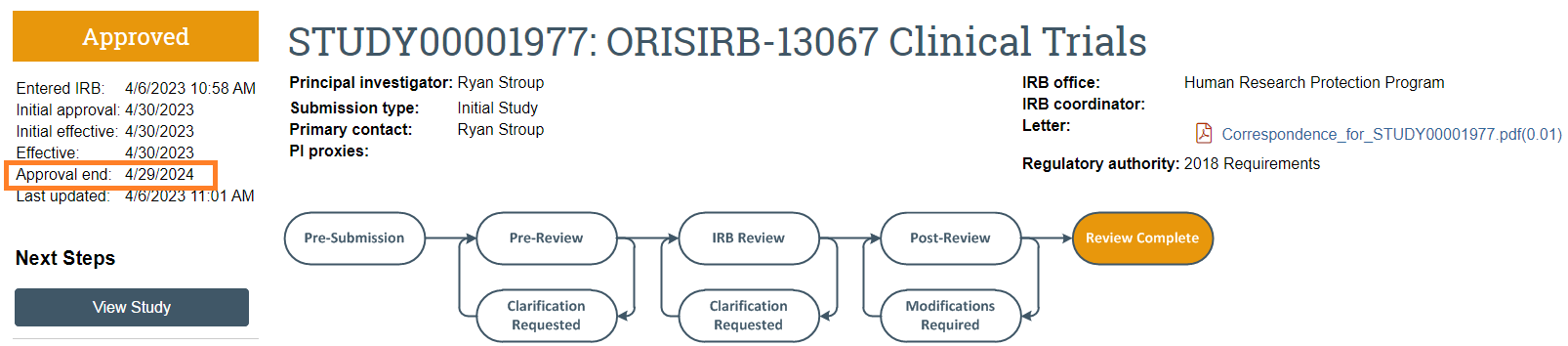
The image below depicts the main study workspace in CATS IRB; "Approval End" indicates that a Continuing Review is required.
Submitting a Continuing Review
Continuing Reviews are submitted to close a study or extend the approval period. Depending on the type of research and research milestones that have been completed, Continuing Review may be performed by expedited review or by full committee review. Follow the steps below outlined below to begin a Continuing Review:
- Login to CATS IRB using your PSU access account.
- Click “IRB” from the top tab menu.
- Select the desired study be clicking on the “Active study” tab and clicking on the study name.
- Click “Create Modification/CR” on the left side of the screen and then select “Continuing Review.” (See image below.)


Note: If any changes are being made, select “Modification and Continuing Review” and refer to the instructions on how to Submit a Modification.
- Complete all questions applicable to Continuing Review.
- Upload any pertinent documents to the Continuing Review form.
- Click “Save and Exit” to return to the Submission workspace where you MUST SELECT “Submit” to send to the IRB for review.
 Only the PI or the PI Proxy can Submit. If Submit is not selected, the continuing review will remain in the pre-submission state and will not be submitted.
Only the PI or the PI Proxy can Submit. If Submit is not selected, the continuing review will remain in the pre-submission state and will not be submitted.
- Once the Continuing Review is submitted, the state will move to “Pre-Review” as pictured below. The PI, PI Proxy, and the Primary Contact will be notified by email when the Continuing Review is approved.