Step 4: After You Submit
Submission Review
After submission, each study is assigned a review level. Studies generally fall into one of three review categories: exempt, expedited, or committee review.
Exempt and expedited studies are reviewed by an experienced IRB Analyst. Committee review level studies go to the IRB committee for review.
The IRB will not approve a submission or make an exempt determination until all study team members have completed the required training in CITI.
Clarifications Requests
IRB Analysts or the IRB Committee may request clarifications or revisions during the pre-review or review of a submission. Such requests will come in the form of a "Clarification Request" action found under the "History" tab of the submission in CATS IRB, pictured below.
Please visit our webpage Respond to Requests for detail instructions on how to perform this function in CATS IRB.
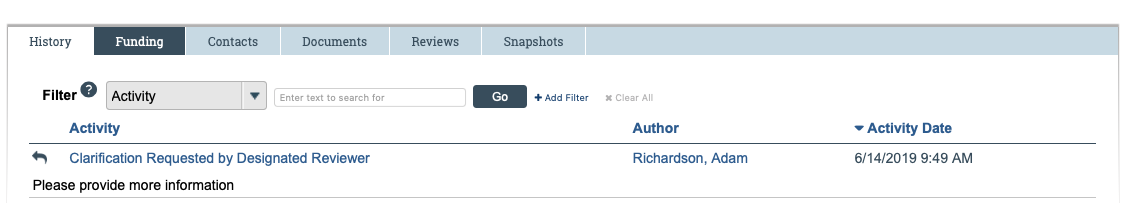
The Clarification Request function is the official communication from the IRB. The study team must respond to requests for clarification within CATS IRB within 30 days. While email exchanges or phone calls with an IRB Analyst may be helpful for understanding clarifications, the study team must submit changes in CATS IRB to fulfill Clarification Requests.
Current Timeline
Currently, the time from submission to pre-review of a submission by an IRB Analyst is approximately 8 business days. Please note that the time to review is not the same as the time to approval. Incomplete submissions or those that do not adhere to IRB policy requirements may require additional time. Review timelines may also be affected by factors outside of the IRB such as other required ancillary reviews, the FDA, Certificates of Confidentiality, etc.
Investigators frequently request that the IRB conduct a priority review. In general, submissions are reviewed in the order in which they are received. The current volume of submissions precludes pulling items out of the queue unless it is a true emergency that will adversely affect study subjects or funding. Reviewing submissions out of order means that other investigator’s submissions will not receive a timely review. Investigators who believe that their submission requires immediate handling, should contact their assigned IRB Analyst and provide justification and a description of the specific circumstances.
Tips to help decrease the time to approval for a submission:
- Start working on the IRB submission well in advance and submit early whenever practicable.
- Provide the appropriate study protocol(s) and protocol site addendum, when applicable, for review, along with all supporting documents (e.g., informed consent forms, recruitment materials, data collections instruments, Sponsor protocol and documentation, full grant proposal, etc.) in CATS IRB and ensure that any discrepancies between these documents have been resolved prior to submission.
- For student research, ensure that the faculty advisor has reviewed the submission for completeness and accuracy prior to submission to the IRB.
- Ensure that all study team members complete their required CITI training prior to submission.
- Provide clear, accurate, and complete responses to IRB requests for clarification (frequently, investigators make changes to the study protocol, but those changes are not carried into supporting documents where necessary).
The HRPP acknowledges that these types of delays interfere with the conduct of research and that many research teams are eager to submit modifications that would allow their research to continue or restart during the pandemic. We are actively working not only to review submissions quickly but also to improve our processes overall. Thank you in advance for your patience.
What's Next?
Move on to Step 5 to learn about managing submissions after IRB Approval, clinical trial requirements, and closing a study.
